Breath volume and measurements of length, area, volume and mass
Created by Dr.Walt Volland, All rights reserved copyright 2005-2013
revised by Dr. Walt Volland September 24, 2013
| You need to post your average personal breath volume early enough so that a class average can be figured from Breath Volume postings in the Breath Volume forum . Your lab report for Dr. Walt Volland's class is completed when you submit your equiz 2 in Canvas |
|
Measurements are a fundamental part of science and technology. The way measurements are made is always on a relative basis. The medieval systems were typically established by royal decree and often referred to the dimensions of body parts. A time line for refinements in the definition of the meter is at |
http://physics.nist.gov/cuu/Units/history.html
|
Another time line is at |
http://lamar.ColoState.edu/~hillger/meter.htm
|
This web site is a source of conversion factors |
http://www.soton.ac.uk/~scp93ch/units/convfact.html
| Units and conversion factors are described and illustrated at these sites |
http://www.wsdot.wa.gov/reference/Metrics/factors.htm
|
This experiment is intended to give you hands on practice with length, area, volume and mass units. You will use the SI units to measure length. You will measure length and width and then calculate area. You will construct a cube with each edge equal to one decimeter to see first hand how the volume of a liter is defined. You will also measure out a liter of water and "feel" the heft of a kilogram. Since water has an approximate density of 1 gram /ml, the mass of the liter of water is essentially 1 kilogram. The actual volume of a container like 2-liter soft drink bottles or 1 gallon plastic jug will be measured. The 2-liter bottle (you may need two of them) or plastic 1 gallon jug will then be used to measure the volume of your exhaled breath. The displacement of water from the container can be done in a sink, but some folks have suggested using a bath tub. I worry about people falling into the tub. You will repeat this measurement three times and report your average value. If you are in Dr. Walt Volland's class post your results in the subject line. Please use the format like this example Duck, Donald -1250 ml , Post the message in the class forum in Canvas under communications at
You will also compare your average value with what your classmates report. You will give reasons why youbelieve there are variations from person to person. You will also make suggestions for possible improvements in the procedure.
|
Supplies and Materials
|
Flexible plastic straw or plastic rubber tubing (available from pet stores, Lowes, Home Depot, etc) Ruler with metric scale showing centimeters and millimeters Measuring cup able to measure milliliters (Pyrex and other "glass" types usually have both metric and American units) One clean 1 gallon plastic jug. Large pan or pot, sink or bath tub where you can invert the water filled 1 gallon jug and blow your breath into the jug. |
READ ALL THE STEPS IN THE PROCEDURE BEFORE STARTING THE EXPERIMENT
Length and area
|
Use the centimeter ruler to measure the length and width of the first page of your textbook in units of centimeters. This page has a list of the element names, symbols and average atomic masses. Record the length and width on the report sheet. Calculate the area for the page to the correct number of significant figures. Record your answer, including the proper units. Convert your area to square millimeters and record your answer. Do the number of significant figures change? |
Volume and the decimeter
|
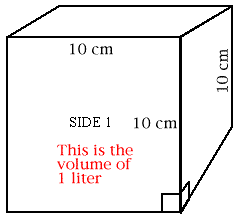
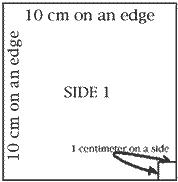
Use your centimeter ruler to draw a line 10 centimeter long. Draw another 10 cm line at a 90 degree angle to the first. Draw two more lines in the same way so you have constructed a square with 10 cm edges. Make a mark 1 cm to the left of the lower right hand corner; make a similar mark 1 cm above the lower right corner. Draw lines 1 cm long perpendicular (at 90 degrees) to the lower and the right edges of your square so you have drawn a small square 1 cm on an edge. Repeat steps 1-4 to make five more squares just like Side 1. Cut out the 6 squares and tape them together to form a cube. Place the little squares at the lower right corner in front and at the upper left rear corner of this cube. The big cube you have built is a cubic decimeter in size. It has a volume of one liter. What is the volume of the little cube whose faces you see in the lower right corner of the big cube? How many of these little cubes fit into the cubic decimeter?
|
|
|
--------------- |
------
------------------ ------ ------ ------ ------ ------ ------
------ |
|
Remember that when length is multiplied by length the new units are length to the power of "2". The unit label has an exponent of "2". This is because the units are squared.
|
|
Remember that when length is multiplied by length is multiplied by length the new units are length to the power of "3". The units are cubed.
|
Mass of one kilogram
|
Gather the 1 gallon bottle or 2 liter bottles whose volume you will measure, and your measuring cup with milliliter calibrations. Be sure your bottles are clean and dry. Pick up the empty bottle and get a sense of its weight. Use your measuring cup to add 1 liter of water to the bottle. Mark the water level with a marking pen. Lift the bottle and its contents. Note the "heft" of the bottle and water. This mass equals a kilogram plus the mass of the bottle. Hopefully this gives you a feeling for a kilogram. |
The volume of a breath of exhaled air
|
Gather the gallon jug, measuring cup, large container, and a flexible straw (18 inches of plastic or rubber tubing available from Petco fish department, Lowe's or Home Depot may be used instead of a straw). Fill the 1 gallon bottle to the very top with tap water. Carefully pour water from the bottle into the measuring cup to determine the volume of water in milliliters that actually fits into the bottle. You will need to transfer a number of portions of water to the measuring cup. Be sure not to fill the cup above its top reference mark. Keep track of the amount of water you pour into the cup. Record the bottle's volume in milliliters to the correct number of significant figures. The bottle will hold more approximately 4 liters. NOTE more than one 2 liter bottle may be needed to collect all of your breath if you do not use a gallon jug. Repeat this step two more times. Then average the 3 values for the volume of your bottle and record the average for the volume of your bottle. Place the pan or other large container in the kitchen sink or bath tub. Add tap water to the pan or pot so you can invert the gallon jug in the pan. it is about half full. Remember to put your pan in the sink or bath tub so the water you blow out of the gallon bottle is contained by the sink or tub. Fill your gallon bottle to the very top. You can seal the bottle by screwing the bottle cap on. Alternately you can place the palm of your hand over the mouth of the bottle to seal the bottle, but this way is more difficult to keep the water in the bottle. Keep your hand tight against the mouth of the bottle and turn the bottle upside down. The water should still be in the bottle because you have the mouth sealed with the lid or your hand. If you screwed the cap on the bottle simply keep the cap on until you are ready to blow into it. Keeping the bottle upside down, carefully put the mouth of the bottle a feel inches below the surface of the water in the large pan or container. You can unscrew the bottle cap to open the bottle. The water should stay in the bottle and not leak out. The water should stay in the bottle as long as the mouth of the bottle stays underwater. Take your flexible straw or tubing and bend it to make a "V" shape. The type of straw is shown here. You will need to adjust the bend to suit your work. The coin (a penny) is included to give an idea of the relative size of the straw. Flexible rubber or plastic tubing simplifies this step. 
Put a short length of tubing inside the mouth of the bottle. You should be able to put your mouth on the other end. Before you put your mouth to the straw, take a deep breath and hold it. Close your lips around the straw and blow out as much air as you can in a steady stream through the straw. Air bubbles will come out the end of the straw and be trapped in the bottle as you exhale. The air will push a volume of water out of the bottle that will equal the volume of the breath you blow into it. This is why you have your large container sitting in the sink! While the mouth of the bottle is still underwater, place the palm of your hand over the mouth to again seal it. Keep the bottle sealed with your hand. Remove the bottle from the container and turn the bottle right side up. Use a measuring cup to measure how much water is needed to bring the water level in your bottle to the top. This gives us an experimental mesure of your breath volume. Record this volume in milliliters. Record the volume of breath you can exhale. Repeat until you have three measurements of the volume of breath you can exhale. Calculate the average volume you can exhale and post your result to the class conference. |
Report sheet for data, results, and answers to questions.
The experiment report for Dr. Volland's class is due midnight of the designated Monday
Use the lab results to complete your equiz 2
|
1. Length and area |
|
Report data with appropriate number of significant figures |
|
1. |
What is the long dimension of the textbook page in centimeters?
|
cm |
2. |
What is the narrow dimension of a textbook in centimeters? |
cm
|
|
3. |
What is the area of a page in square centimeters? set up for calculation below and the answer with units in the box to the right
|
|
|
4. |
What is the area of a page in square millimeters? set up for calculation below and the answer in the box to the right.(This is a second order conversion, exponent is a "2" not a "1" on the units.)
|
mm2 |
|
2. Volume and the decimeter |
|
When you constructed your cubic decimeter, you were to mark the outline of a smaller cube on the lower right hand corner. What is the volume of the little cube? |
|
How many of these little cubes will fit into the cubic decimeter? |
|
Show any calculations to support your answer here.
|
|
3. Mass of one kilogram. |
|
Does the "heft" or heaviness of the mass of a kilogram surprise you? Briefly explain your answer.
|
|
Name any object that you handle regularly that has a similar mass and "heft".
|
|
4. Your breath volume of exhaled air from the volume of water needed to fill bottle to the top after exhaling into it. |
|
Volume of water in milliliters (ml) to fill bottle to its top |
|
Trial 1 |
|
|
Trial 2 |
|
|
Trial 3 |
|
|
Average volume of water needed to refill the bottle |
|
|
Volume of your breath calculated measuring the volume of water needed to refill the bottle after blowing into it. breath volume = volume water added to bottle to refill it. Example for three trials
|
|
Record the volume of breath you can exhale below. Post your breath volume with your name and average volume in ml in the subject line at the appropriate Canvas discussion forum for your class section. Post your average volume of breath in ml in the following format Clinton, Hillary-2800 ml Lincoln, Abraham-3400 ml Duck, Donald-1100 ml Duck, Daffy-1400 ml This allows people to see the value without opening the message and saves time. Use the Breath volume forum part of the communications area for Canvas. The current forum is titled
Use five values posted by the other members of the class to calculate the average breath volume for the class in ml. Show the values you used to calculate your average. Report the average to the proper number of significant figures. Please do not give 8 digits just because your calculator generates them. Round off to the appropriate average. Example: 1100 ml + 1560 ml + 1930 ml = 4590 ml total The average volume is 1530 ml which should be rounded off to 1500 ml . There is a consistent value in the "thousands" place. Variations begin in the "hundreds" place. This has only two significant figures. The uncertainty or variation starts in the hundreds place. All the digits in the "ones" and "tens" places are not meaningful and need to be dropped |
|
|
|
Do not use your own volume to figure the class average.
Use volumes posted in the Canvas Breath forum
|
Student 1 |
|
|
Student 2 |
|
|
Student 3 |
|
|
Student 4 |
|
|
Student 5 |
|
|
Class average breath volume for six class readings in ml |
ml |
|
What is the difference between the class average (five values) and your measured volume. Give difference in milliliters and liters. difference in milliliters difference in liters. |
|
|
|
What are these differences as a percent of your volume? Remember percent = 100 x ( difference in volumes / average volume) Does the percent difference change when you change units? Show your set up for calculations. |
|
Give a suggestion on how you think the experiment could be improved. Identify some aspect you liked about the experiment. Identify something in the experiment that was new to you.
|
revised by Dr. Walt Volland September 24, 2013
The experiment report is due for midnight of the designated Monday